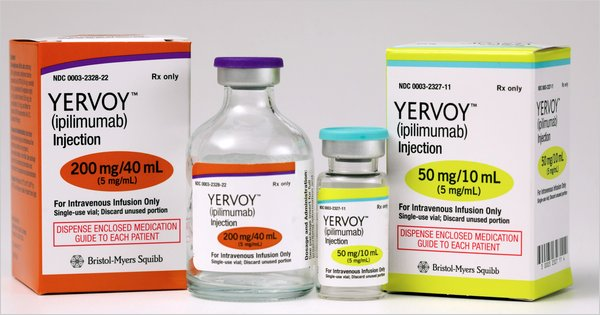
What is ipilimumab 50?
Ipilimumab is a generic medicine classified under monoclonal antibodies. In 2011, the US Food and Drug Administration (FDA) has approved Ipilimumab for treating advanced-stage Melanoma. Melanoma is deadly skin cancer. Later, the European medicines agency also approved this medicine.
Treatment
Initially, the doctor only prescribes Ipilimumab when Melanoma spreads or cannot be removed by the surgery. But currently, it was approved for the stage 3 patients as adjuvant therapy. Ipilimumab is safe for both children and adult patients. Some doctors prescribe ipilimumab, as a generic name or some may refer to trade medicine of ipilimumab i.e. Yervoy.
Dosing
Two doses of ipilimumab are available i.e. 50mg and 200mg. Therefore, ipilimumab 50 means 50 mg vial (5 mg/mL). The medicine is given to the patients through infusion by using an IV line and takes time over 90 minutes.
Storage indication
Storage indication: The FDA approved label mentioned that ipilimumab medicine should store under refrigeration at 2°C to 8°C (36°F to 46°F). Do not freeze the ipilimumab. Vials should be kept stored in a dark place and protected from light.
Side effects
Ipilimumab has some side effects that are commonly noticeable. Rash, pruritus, fatigue, colitis, and diarrhea, etc. are common side effects. However, dosing at the 10 mg/kg dose includes headache, insomnia, nausea, vomiting, weight loss, pyrexia, and decrease appetite.
How does Ipilimumab work?
Our immune system contains T-cells that fight against infections and diseases. The T cells have the ability to detect and destroy melanoma cells. But T-cell surface containing CTLA-4, a protein can stop T-cell’s action. Ipilimumab attaches with CTLA-4 protein and destroys them so that T-cells can destroy melanoma cells.
Ipilimumab activates Cytotoxic T lymphocytes activity that can recognize and destroy the cancerous cells. Inhibition of Cytotoxic T lymphocytes activity can alter this mechanism. Ipilimumab boosts the immune response against cancerous cells.
Indication
Ipilimumab is a prescribed medicine used to treat melanoma, a kind of skin cancer.
- When melanoma cannot be removed through surgery but spreading to other body parts
- To prevent relapsing of melanoma
Availability in India
Still now, Ipilimumab is not available in India. But patients who have import permission from the Indian Government can access the product.
Yervoy is the tread name of ipilimumab. Both 50 mg and 200 mg. are supplied for Tenders, Name patient medicine, Orphan drug, Emergency imports, Reference listed drugs, Comparator Drug, and Innovator samples, etc.